Areas of Study
Ca2+ homeostasis and animal behavior
Education
- University of Utah2007Post doctoral research
- University of British Columbia2002Post doctoral research
- University of British Columbia2000PhD
Research
Research in our lab focuses on the genetic pathways and molecular mechanism that regulate Ca2+ homeostasis and animal behavior. Ca2+ is a critical mediator of many cellular processes, including muscle contraction, mitochondrial activity, transcription, cell division, and synaptic vesicle release. Paradoxically, Ca2+ can also trigger cell death and cellular necrosis. Therefore, dysregulation in Ca2+ signaling can disrupt cellular homeostasis. Consequently, Ca2+ levels need to be tightly controlled. Indeed, defective calcium signaling has been implicated in many neurodegenerative diseases, muscular dystrophies and heart disease. Since cellular signaling mechanisms are largely phylogenetically conserved, our studies utilize the free-living nematode Caenorhabditis elegans, a model system that provides exceptional experimental advantages for describing fundamental biological processes. These advantages include genetic tractability, small nervous system with mapped neuronal connectivity, transparent (individual cells can be visualized in vivo), stereotyped behaviors, and efficient methods for studying large numbers of animals. Our research employs in vivo Ca2+ signaling in concert with genetic, molecular and cell biological techniques to explore organismal behavior. Listed below are the current specific areas of research in the lab:
We have recently demonstrated that SEL-12, the C. elegans presenilin ortholog, has a role in mediating endoplasmic reticulum-mitochondrial Ca2+ homeostasis. Disruption of presenilin function results in an increase in mitochondrial Ca2+ levels and alters mitochondrial metabolism promoting protein homeostasis collapse and neurodegeneration. Presenilin is a highly conserved protein found from plants to humans that is extensively found on endomembrane structures (e.g., endoplasmic reticulum and lysosome) of most cell types. However, the role presenilin has in the endomembrane system is not clear. Importantly, mutations in human presenilin are the most common cause of early onset familial Alzheimer’s disease. Despite the identification of the involvement of presenilin in Alzheimer’s disease over 20 years ago, the functional consequences of mutations in presenilin causing Alzheimer’s disease are not understood. Thus, we are employing C. elegans to determine the role presenilin has in mediating mitochondrial Ca2+ homeostasis.
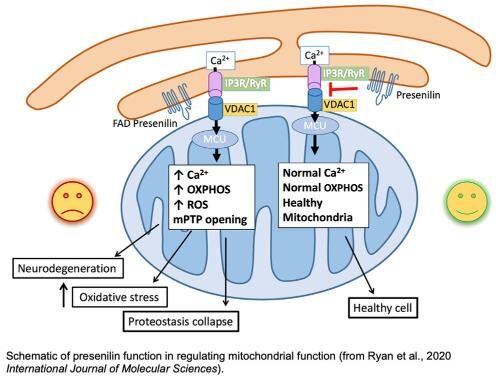
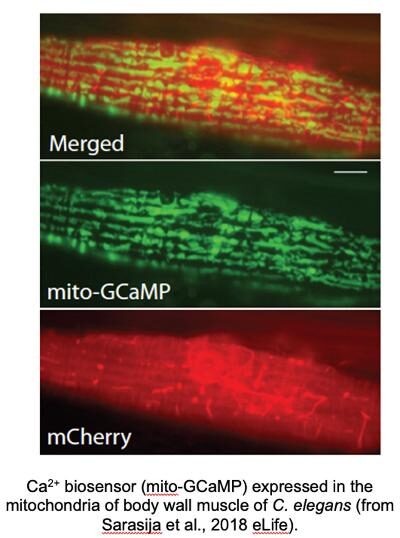
Since we have found a fundamental role of mitochondrial Ca2+ homeostasis in mediating neuronal health, we are investigating the mitochondrial Ca2+ uniporter complex (MCUcx). Ca2+ uptake into the mitochondria is mediated by the evolutionarily conserved mitochondrial Ca2+ uniporter complex (MCUcx). The MCUcx is composed of four core components: the pore forming MCU protein, an auxiliary subunit EMRE (essential for MCU regulator) and the gatekeepers MICU1 and MICU2/3. With the recent molecular identification of MCUcx components, many current studies have implicated altered mitochondrial Ca2+homeostasis as having a fundamental role in promoting neurodegeneration. Thus, we are exploiting the genetic amenability and simplicity of C. elegans to interrogate the role of the four core components of the MCUcx to establish a foundation of MCUcx function in an in vivo model system and to discover new gene products that are involved in mitochondrial calcium influx.
Publications
Laboy JT, Bonner J, Norman KR. DEC-7/SUSD2, a sushi domain-containing protein, regulates an ultradian behavior mediated by intestinal epithelial Ca2+ oscillations in Caenorhabditis elegans. Am J Physiol Cell Physiol. 2023 May 1;324(5):C1158-C1170. doi: 10.1152/ajpcell.00552.2022.
Ryan KC, Laboy JT, Norman KR. Deregulation of Mitochondrial Calcium Handling Due to Presenilin Loss Disrupts Redox Homeostasis and Promotes Neuronal Dysfunction. Antioxidants (Basel). 2022 Aug 24;11(9):1642. doi: 10.3390/antiox11091642.
Ryan KC, Ashkavand Z, Sarasija S, Laboy JT, Samarakoon R, Norman KR. Increased mitochondrial calcium uptake and concomitant mitochondrial activity by presenilin loss promotes mTORC1 signaling to drive neurodegeneration. Aging Cell 2021 20(10): e13472.
Ryan KC, Ashkavand Z, Norman KR. The Role of Mitochondrial Calcium Homeostasis in Alzheimer's and Related Diseases. Int J Mol Sci 2020 21(23):9153.
Nallanthighal S, Rada M, Heiserman JP, Cha J, Sage J, Zhou B, Yang W, Hu Y, Korgaonkar C, Hanos CT, Ashkavand Z, Norman K, Orsulic S, Cheon DJ. Inhibition of collagen XI alpha 1-induced fatty acid oxidation triggers apoptotic cell death in cisplatin-resistant ovarian cancer. Cell Death Dis 2020 11(4): 258
View Kenneth R. Norman's articles on the National Institute of Health's PubMed website.
PDFs
Corrupted ER‐mitochondrial calcium homeostasis promotes the collapse of proteostasis
Measurement of Oxygen Consumption Rates in Intact Caenorhabditis elegans
Role of Presenilin in Mitochondrial Oxidative Stress and Neurodegeneration in Caenorhabditis elegans
Analysis of Mitochondrial Structure in the Body Wall Muscle of Caenorhabditis elegans
